

Most of the modern explosives are based on TriniTroToluene (TNT) explosive. In the figure Improvised Explosive Devices (IED) loaded with TNT.
The technology for revealing explosives is based on the detection of molecules of explosives released in the air by means of supersensitive sensors based on properly functionalised nanostructures.
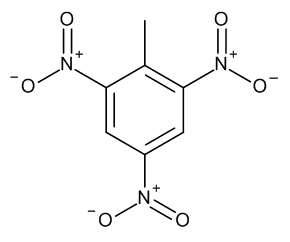
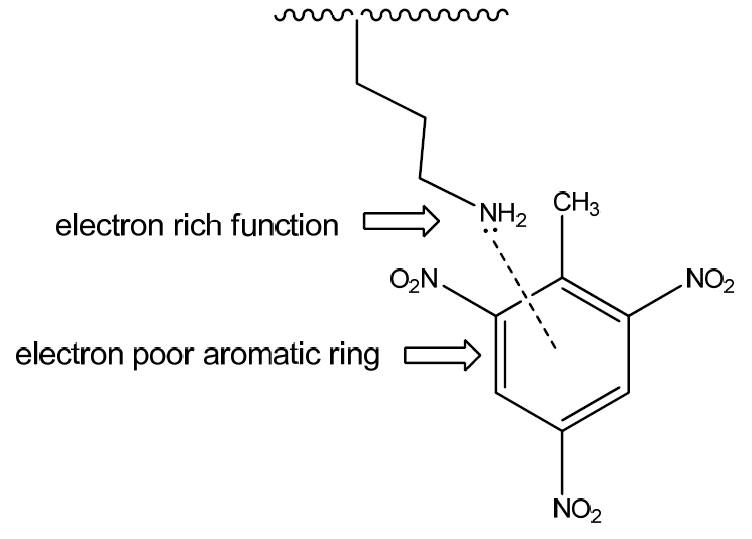
The recognition of explosive molecules are performed by the strong attractive acid-base pairing interaction between the electron poor aromatic ring of TNT molecule and electron rich amino groups NH2:
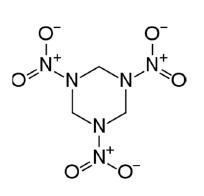
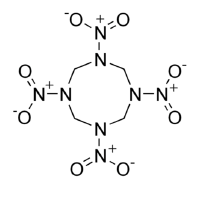
Analogous interactions are expected with other explosives like RDX (1,3,5-trinitroperhydro-1,3,5-triazine) and HMX (octahydro-1,3,5,7-tetranitro-1,3,5,7- tetrazocine) which are the most common modern explosives
Explosive molecules are attracted by amino groups attached to carbon nanotubes or germanium nanowires terminating with amino groups after a process called functionalization.
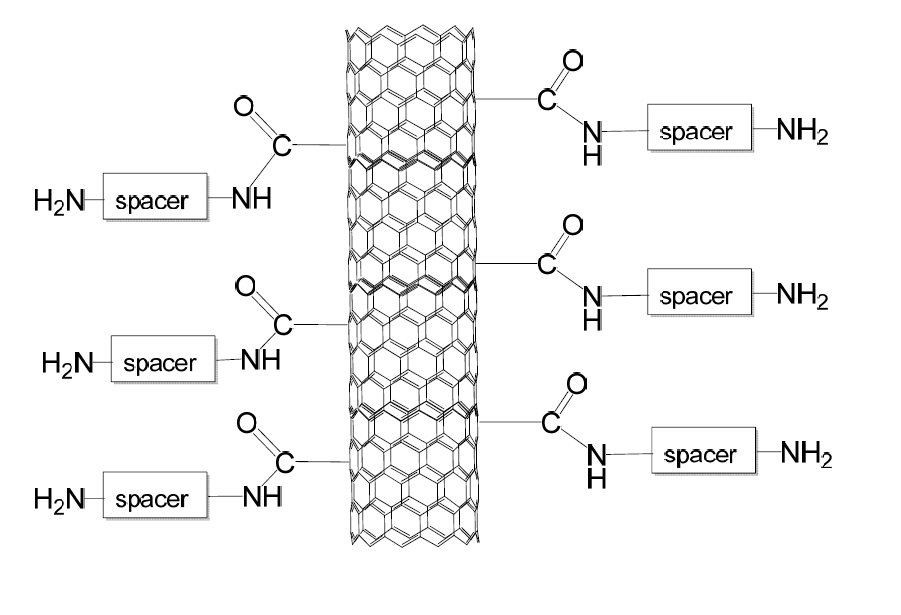
Carbon nanotube after functionalization terminating with NH2 amino-groups

Germanium nanowires before the functionalization process
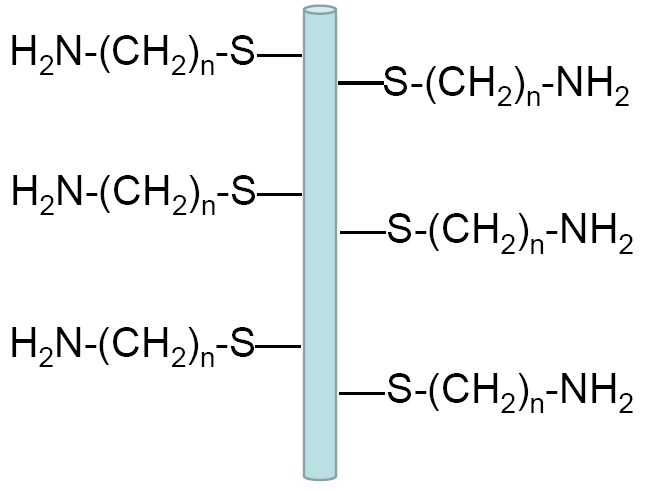
Germanium nanowire after functionalization terminating with amino-groups
